Anaerobic Respiration
Anaerobic Respiration in Eukaryotes
- When life began, there was no oxygen so aerobic respiration was not possible.
- Anaerobic respiration has a net production of two molecules of ATP per glucose molecule.
- In eukaryotic cells, anaerobic respiration is now used as an emergency measure to keep vital processes functioning.
- Organisms can be classified based on their dependance of oxygen...
- Obligate Anaerobes; Cannot survive in the presence of oxygen. Almost exclusive to prokaryotes.
- Facultative Anaerobes; Synthesise ATP by aerobic respiration is possible, but can switch to anaerobic respiration is necessary. For example, yeast.
- Obligate Aerobes; Can only synthesise ATP in the presence of oxygen. Some individual cells in obligate anaerobes can be classed as facultative anaerobes, but this is only temporarily as it produces compounds which need to be broken down when oxygen is available again.
- The process in which organic compounds are broken down into simpler inorganic compounds are without the use of oxygen or an electron transport chain.
- As glucose is not broken down as much as it is in aerobic respiration, only a small quantity of ATP is produced, by substrate level phosphorylation alone.
- The synthesis of ATP by chemiosmosis stops when there is no oxygen to accept protons and electrons at the end of oxidative phosphorylation.
- As the flow of electrons stops, NADH and FADH2 cannot be oxidised as there is nowhere for the electrons to go
- This means NAD and FAD are not regenerated.
- This means the decarboxylation and oxidation of pyruvate and the Krebs cycle stops, as no coenzymes are available to remove the hydrogen ions.
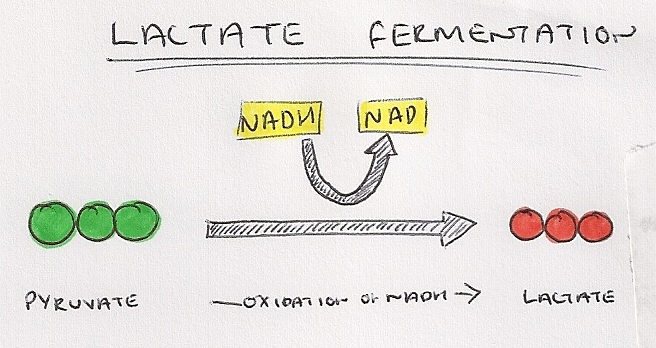
- Lactate fermentation produces lactate, and occurs in animal cells.
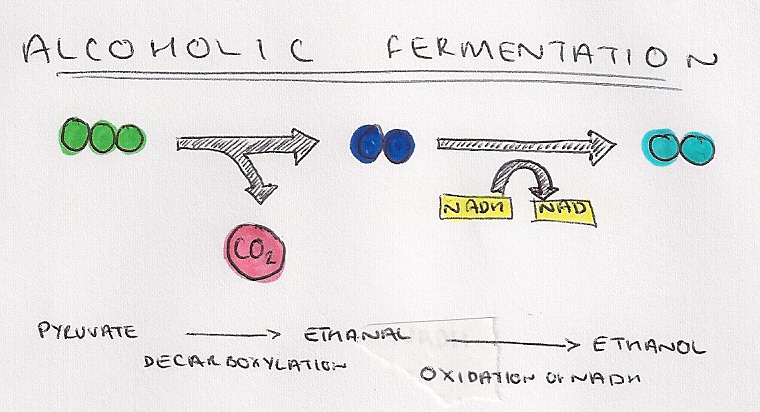
- Alcoholic fermentation produces ethanol and CO2, and occurs in yeast and some plant cells.
- Pyruvate can act as a hydrogen acceptor, taking hydrogen ions from NADH, catalysed by the enzyme lactate dehydrogenase.
- Pyruvate is converted into lactate (lactic acid) and NAD is regenerated.
- Lactic acid is converted back into glucose in the liver but oxygen is required to complete the process, which is the reason for the oxygen debt after exercise.
- This can be used to keep glycolysis going to allows for a small amount of ATP synthesis.
- Lactate fermentation cannot happen indefinitely because...
- The reduced quantity of ATP produced would not be enough to sustain vital processes over time.
- Accumulation of lactic acid caused a fall in pH (making it more acidic) leading to denaturation of proteins.
- E.G. respiratory enzymes and muscle fibres would cease to function.
Alcoholic Fermentation in Yeast and many Plants
- Unlike lactate fermentation, alcoholic fermentation is not a reversible process.
- Pyruvate is converted to ethanal by decarboxylation, catalysed by the enzyme pyruvate decarboxylase.
- Ethanal then accepts a hydrogen atom from NADH, producing ethanol.
- This process regenerates NAD and allows for the continuation of glycolysis.
- Alcoholic fermentation is a long term process which can continue indefinitely in the absence of oxygen.
- However, ethanol is a toxic waste product to yeast cells, which can kill them if it accumulates to more than 15%
- This is allowed to happen in brewing.
Investigation into Respiration Rates of Yeast
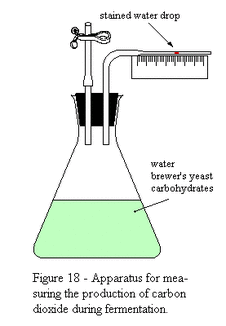
- The apparatus shown could be used to measure the rate of carbon dioxide production of a yeast suspension.
- The glucose solution provides a respiratory substrate, and the flask is sealed to ensure anaerobic conditions.
- As yeast respires, an increasing volume of CO2 in the flask is produced, increasing pressure and causing the stained water drop to move along the tube.
- The distance moved by the water drop can be used to calculate the increase in volume of CO2 over time, to measure respiration.
- As respiration is not 100% efficient, energy is lost as heat when organisms respire.
- Sensors and a data logger can be used to measure the temperature increase of a respiring yeast solution. The increase in temperature can be used to calculate the rate of respiration, and be graphically represented.
Large and Small Scale Adaptations to Low Oxygen Environments
- Bacterial Adaptation
- Different groups of bacteria have evolved to use nitrate ions, sulphate ions or carbon dioxide as a final electron acceptors in low oxygen environments, enabling them to live in anaerobic conditions.
- E.G. Methanogens: an anaerobic bacteria present in the digestive system of ruminants such as cows, which digest cellulose from grass cell walls into products which can be further digested. The final electron acceptor in methanogens respiratory pathway is CO2, producing methane and water.
- Mammalian Adaptation
- Marine mammals that dive for long periods of time have a range of adaptations for surviving when they cannot take in extra oxygen.
- Biochemical Adaptations
- Greater concentrations of haemoglobin and myoglobin maximises oxygen stores in muscles used for swimming, delaying onset of anaerobic respiration.
- They also have a greater tolerance for lactic acid and carbon dioxide.
- Physiological Adaptations
- A modified circulatory system means they can perform peripheral vasoconstriction, prioritising blood flow to the heart, brain and muscles for more efficient oxygen use
- They can slow the heart by 85%, resulting in bradycardia, reducing energy demand.
- They can exchange 85-95% of the air in their lungs, allowing for far more oxygen intake each breath. (Humans only exchange 15% each breath).
- Physical Adaptations
- Streamlining of the body reduces water resistance, therefore saving energy when swimming.
- Fin-shaped limbs allow for more efficient propulsion, therefore saving energy when swimming.
- Biochemical Adaptations
- Marine mammals that dive for long periods of time have a range of adaptations for surviving when they cannot take in extra oxygen.