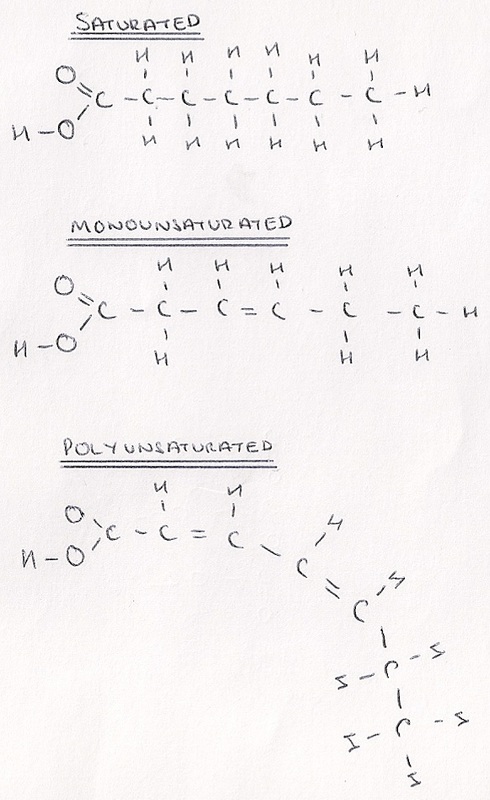Lipids
Lipids:
- aka fats and oils.
- Made of Carbon, Hydrogen and oxygen.
- They are complex, large molecules called macromolecules.
- Not water soluble.
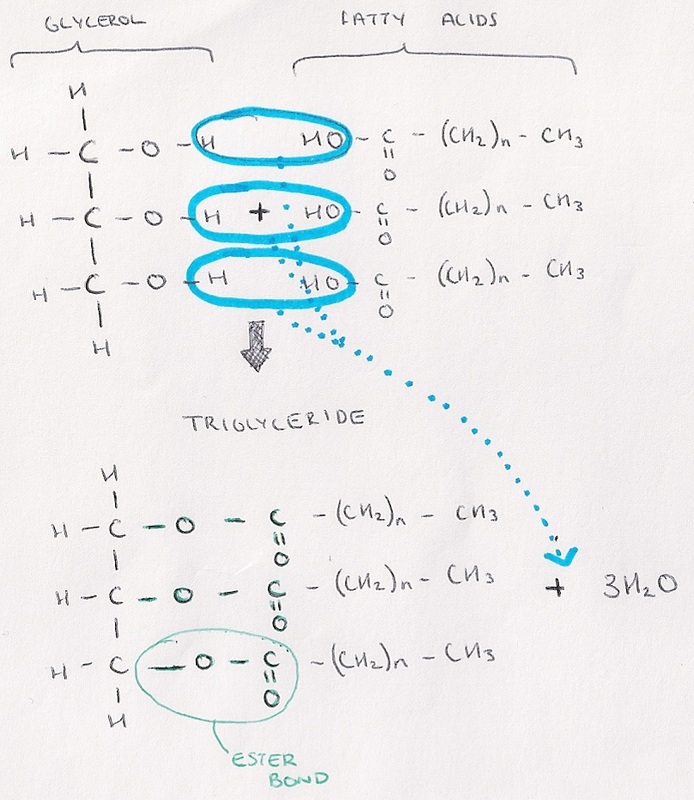
- Made by combining a glycerol molecule with three fatty acid chains.
- Glycerol is an alcohol.
- Fatty acids are carboxylic acids, because they have a carboxyl groups and a hydrocarbon chain.
- The bond between glycerol and fatty acid chains is called and ester bond, and is formed in esterification (a type of condensation reaction.
- Triglyceride are broken down into their constituent parts by resupplying three water molecules in a hydrolysis reaction.
- Saturated fatty acid chains have no double bonds between carbon atoms.
- Mono-Unsaturated fatty acid chains have one double bond between carbons in the entire chain.
- Polyunsaturated fatty acids have multiple double bonds between carbon atoms in the chain.
- Polyunsaturates bend and kink so cannot be packed tightly, making them liquid at room temperature.
- Plants generally contain unsaturated lipids, whereas humans contain saturated lipids.
Phospholipids:
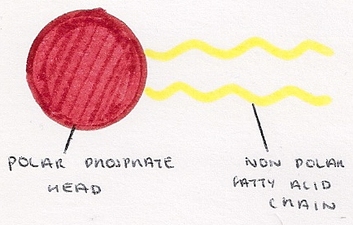
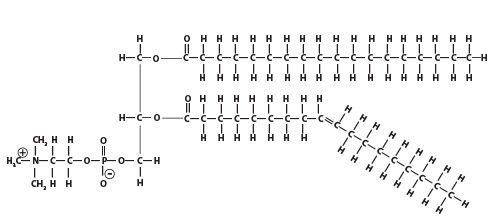
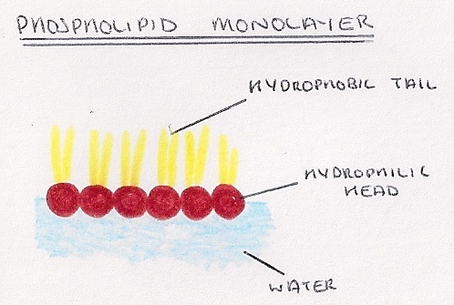
- Modified triglycerides which contain a phosphate head instead of a third fatty acid chain.
- Phosphate head is charged and so is soluble in water.
- It is hydrophilic and interacts energetically favourably with water.
- Fatty acid tails are not charge so are insoluble in water.
- They are hydrophobic and do not react energetically favourably with water.
- As a result of their hydrophilic/hydrophobic structure they will form a layer on the surface of water, with the phosphate heads in the water, and the tails sticking out; Because of this they are called surfactants (surface active agents).
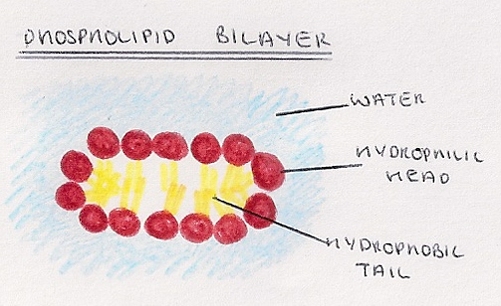
- They can also form a phospholipid bilayer when surrounded by water, with the heads facing out and the tail facing inwards.
- They play a key role in forming cell membranes.
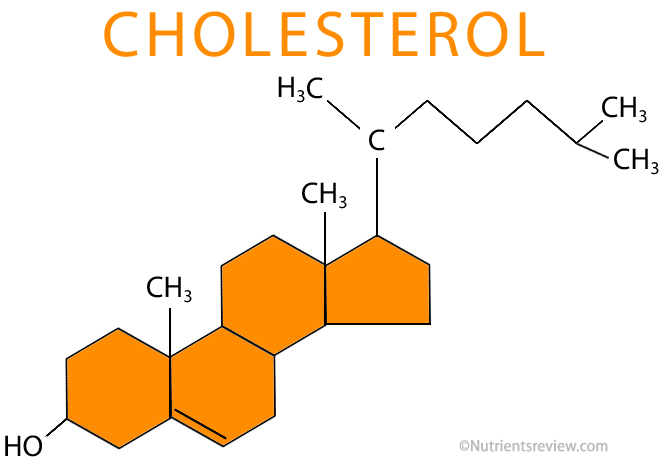
Sterols:
- Steroid alcohols.
- They are lipids, but neither fats nor oils and have little in common with them structurally.
- They are complex alcohols with a four carbon ring structure and a hydroxyl group at one end.
- The hydroxyl group is polar, but the carbon rings aren't and this means the molecule has a hydrophobic/hydrophilic structure, and share many characteristics with phospholipids.
- Cholesterol is a sterol which is important in maintaining the stability of cell membranes.
- Cholesterol also manufactures vitamin D, steroid hormones and bile.
Roles of Lipids:
- Membrane formation
- Hormone production
- Electrical insulation for nerve impulse transmission
- Waterproofing
- Thermal insulation
- Cushioning
- Buoyancy
Identification of lipids:
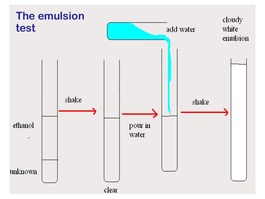
- The presence of lipids can be identified by doing an emulsion test.
- Mix sample with ethanol
- Mix sample with water
- Shake
- If a milky white emulsion forms, then a lipid must be present.
Changing Health Advice:
- Hard to get definitive health advice because
- Not enough tests have been done.
- Hard to isolate the effect of single factors such a amount of lipids.
- Can be hard to measure in some cases.
- New technological advances give us better insights into how the body works.
- Data is often flawed by generalisability issues.
- Headlines in tabloids often cherry-pick data from studies giving a skewed representation, just to be eye-catching.
- Unsaturated fats have a kink in the chain so they are less dense as they cannot be so tightly packed together, so are generally healthier.
- Scientists aimed to make a healthier version of butter from plant oils, by hydrogenating the fat or removing the double bonds, making the oil solid.
- An unwanted byproduct of this process was the creation of trans fats - where oils have become saturated.
- Trans fats increase shelf life, but have also been linked to heart disease.