Structure of Proteins
Key Terms:
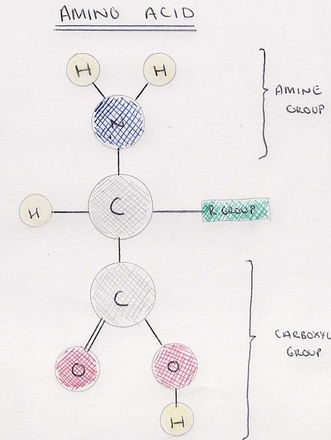
Amino Acid: a monomer used to build polypeptides (and therefore proteins).
Peptide: polymers made of multiple amino acid molecules.
Protein: Biological macromolecules made of multiple polypeptides.
Amino Acids:
- All amino acids have the same basic structure.
- Variable R-Groups are what make amino acids different to each other.
- There are 20 different amino acids found in cells.
- 5 are non-essential, as our body makes them from other amino acids.
- 9 are essential, and can only be obtained from what we eat.
- 6 are conditionally essential, as they are only needed in growing children.
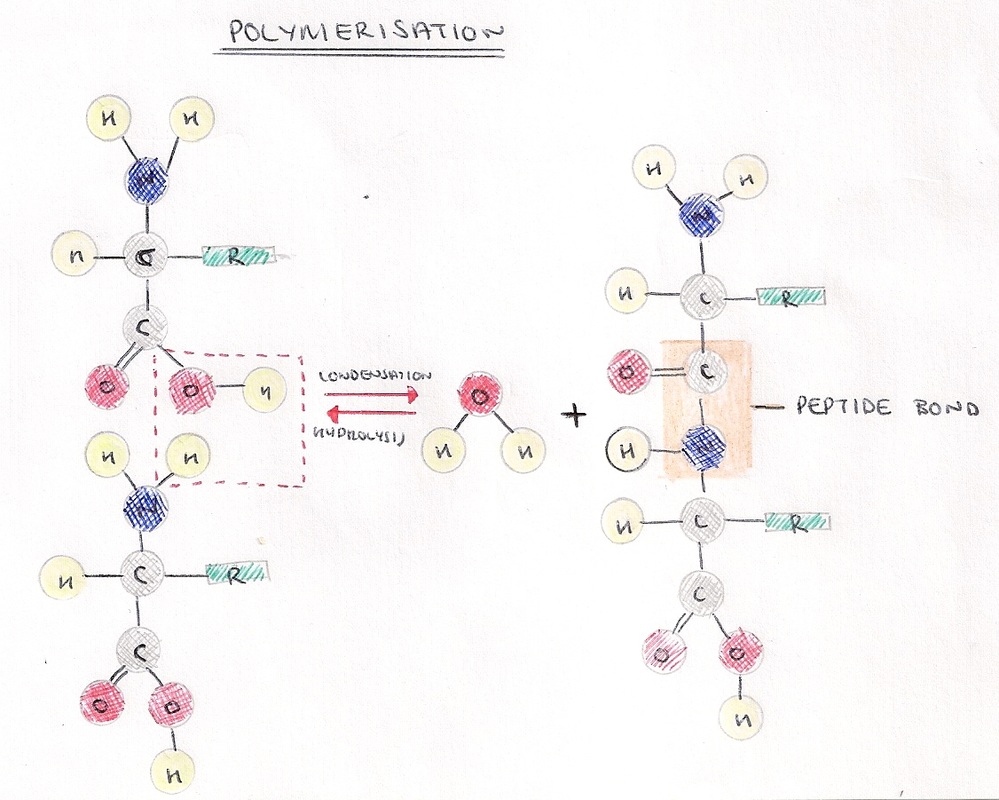
- Amino acids join when the amine and carboxylic groups of two amino acids react.
- A peptide bond is formed in a condensation reaction.
- Two amino acids joined together form a dipeptide. Any more makes a polypeptide.
- The synthesis of peptides is catalysed by peptidyl transferase in ribosomes.
- Interaction between the R groups of amino acids in polypeptides forms different bonds, which cause the protein to fold into different shapes.
Thin Layer Chromatography:
Primary Structure
- A technique used to separate the individual components of a mixture, or example amino acids in a substance.
- Stationary Phase
- Adhesive is applied to rigid surface
- Amino acids are then added to one layer of the adhesive
- Amino acid end is submerged in an organic solvent
- Mobile Phase
- After being dipped in solvent, the amino acids move up the adhesive
- The rate at which the amino acids move through the adhesive depends on the hydrogen bonds they have with the adhesive and their solubility in the solvent.
- Different amino acids will move different distances in the same amount of time, separating them out form each other.
- Pencil line drawn 2cm away from the bottom of the chromatography plate.
- Equally spaced points are marked along the line in pencil.
- Amino acid solution is dabbed onto each point using a capillary tube, labelled and allowed to dry.
- Chromatography plate is then placed in a solvent, and left until the solvent had reached almost to the top of the plate, and then allowed to dry.
- Ninhydrin spray was sprayed on the chromatography plate, causing the amino acids to react with the spray and produce purple spots where the amino acids were.
- Retention factor (Rf) = distance travelled by component ÷ distance travelled by solvent. By comparing these result to a database the amino acids can be identified.
Primary Structure
- The sequence in which amino acids join to form a polypeptide.
- Directly coded for by the DNA.
- The primary structure ultimately affects what shape the protein will fold into because on the interactions between r groups.
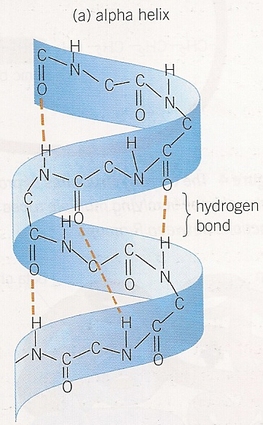
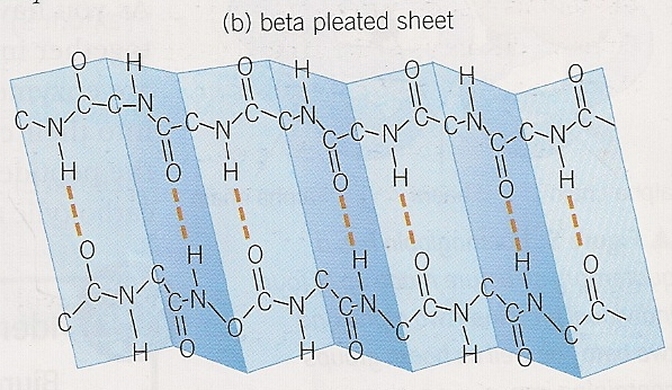
- Only bonds involved are peptide bonds.
- Oxygen, hydrogen, and nitrogen atoms interact, coiling the polypeptide into an alpha helix or folding it into a beta pleated sheet.
- Result of hydrogen bonding.
Tertiary Structure:
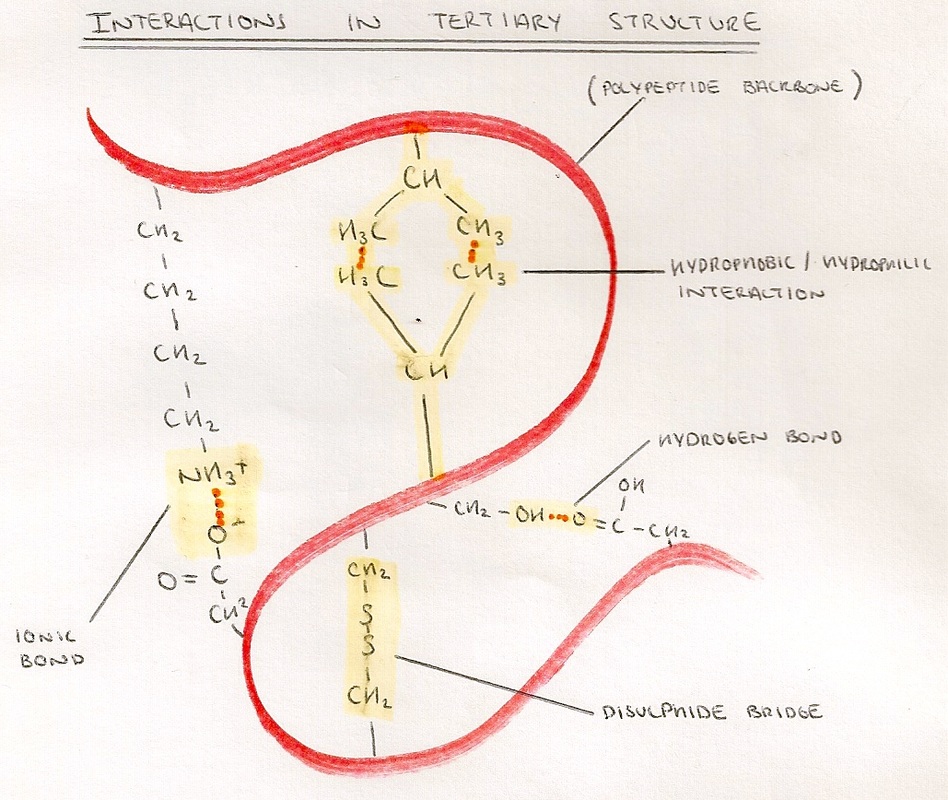
- This is the folding of the protein into its final shape.
- The R Groups of the amino acids are brought close enough to interact by the coiling or folding of secondary structure.
- Bonding includes:
- Hydrophobic/Hydrophilic interactions - weak interactions between polar and non-polar R-groups.
- Hydrogen Bonds - weakest bonds formed between hydrogen and other elements.
- Ionic Bonds - stronger bonds that form between oppositely charged R-groups.
- Disulphide Bridges - strongest bonds which form between R-Groups which have sulphur in.
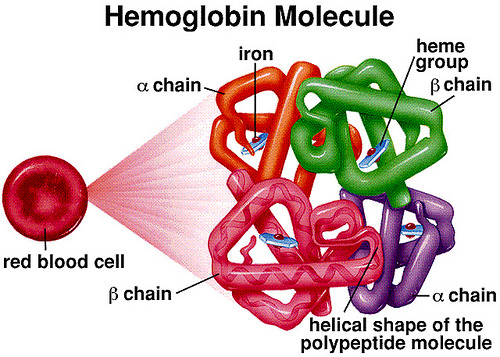
Quarternary Structure:
- Comes from the association of multiple proteins called subunits.
- Bonds/Interactions are the same as in tertiary structure, but between subunits.
- Can also include the addition of a prosthetic group such as haem, in haemoglobin.
Breakdown of Peptides:
- Proteases are enzymes which catalyse the breakdown of polypeptide chains into their constituent amino acids, using a water molecule in a hydrolytic reaction.
- The biuret test can be used to identify the presence of proteins.
- Liquid sample is mixed with equal volume of 10% sodium hydroxide solution.
- 1% copper sulphate solution is added a few drops at a time until the sample turns blue.
- Mix solution ad leave for 5 minutes.
- If proteins are present, the peptide bonds form violet coloured complexes with copper ions.